Marble Chips And Hydrochloric Acid Graph

Investigating the rate of reaction between marble chips calcium carbonate and hydrochloric acid aim.
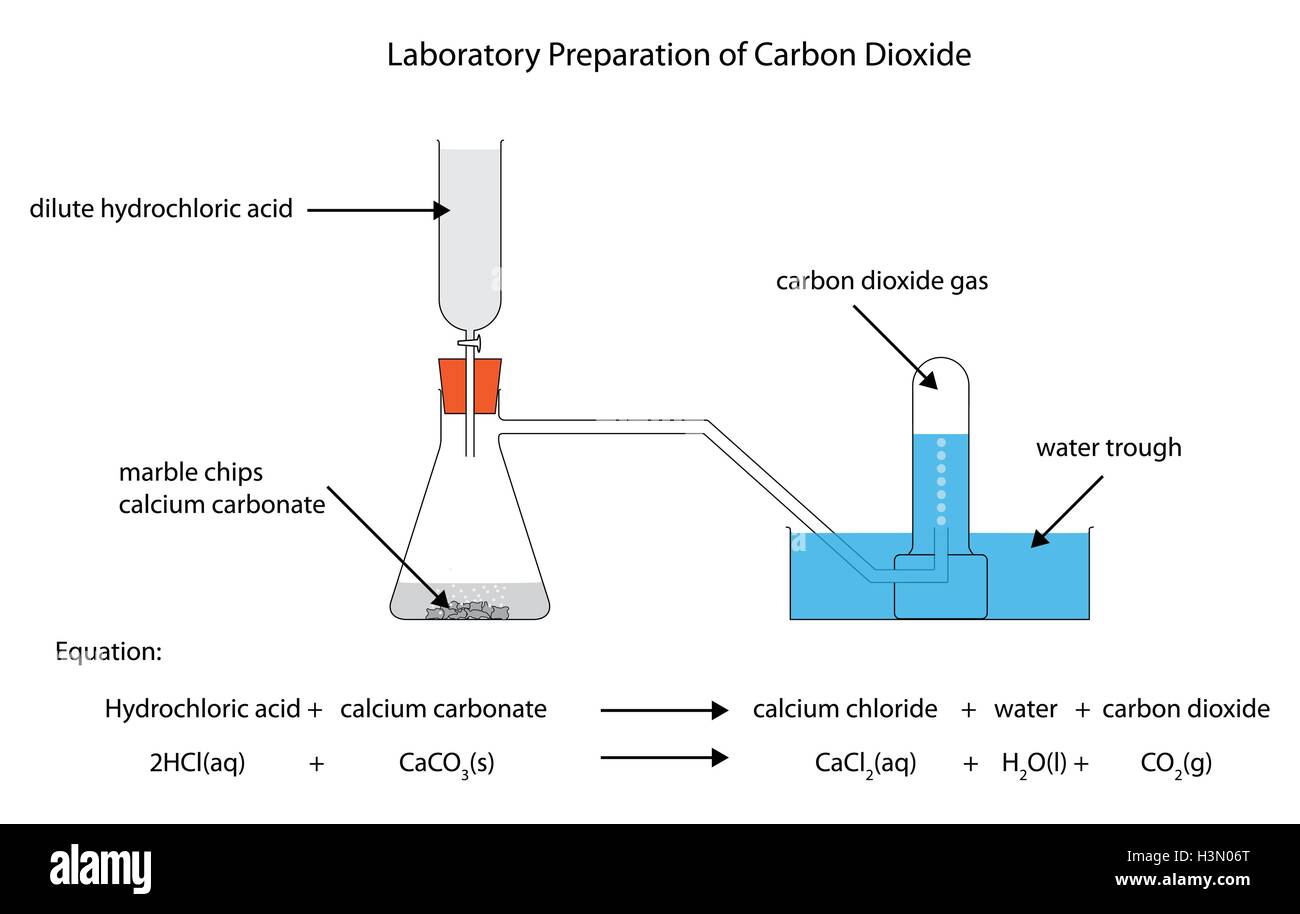
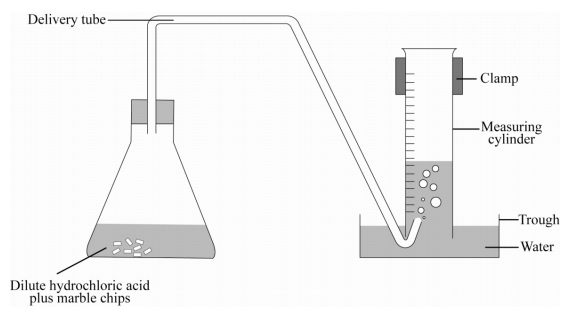
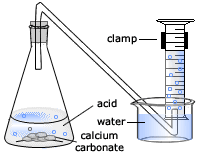
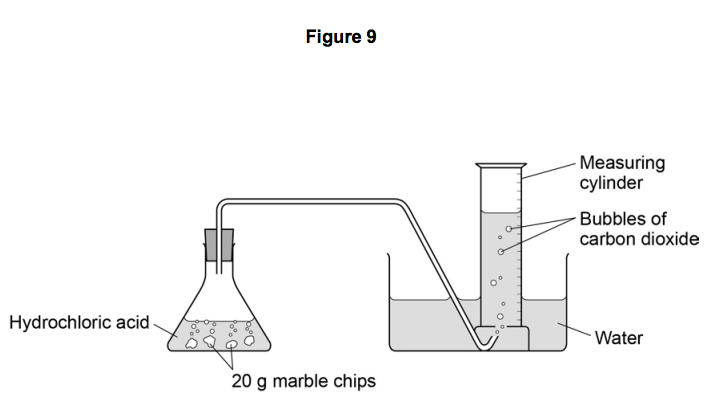
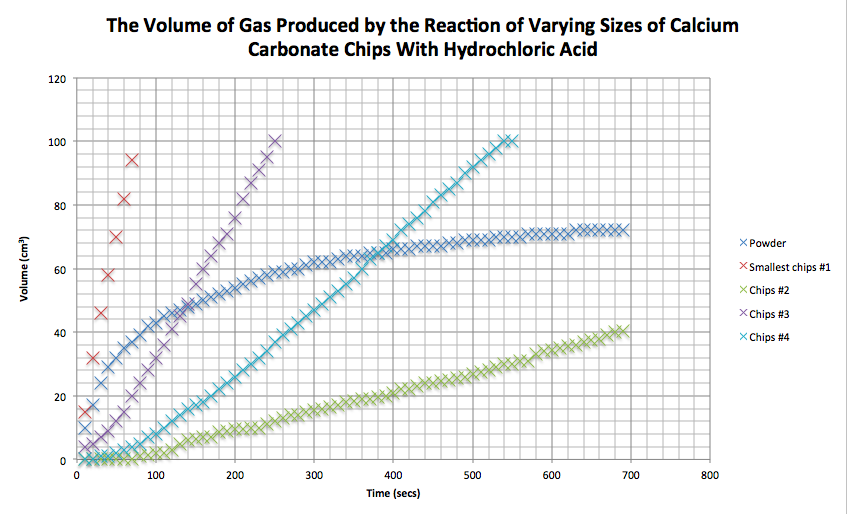
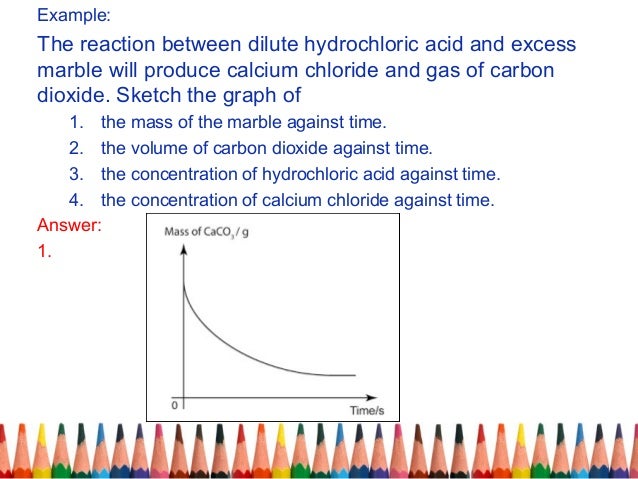
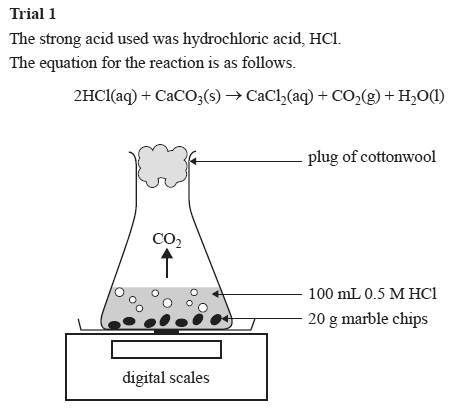
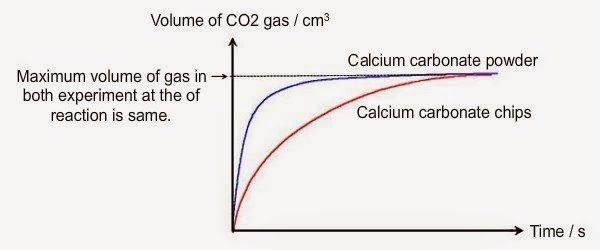
Marble chips and hydrochloric acid graph. The rate of this reaction can be changed by changing the size of the marble chips. A tube to connect the conical flask to the measuring cylinder. Conical flask glass jam jars measuring cylinder stop clock watch with seconds stop watch app direct reading balance dilute hydrochloric acid cotton wool marble chips method. Place 40cm 3 of hydrochloric acid in an conical flask.
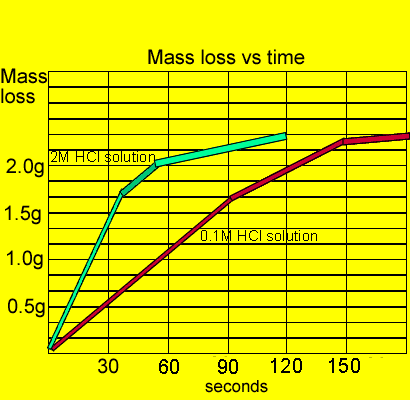
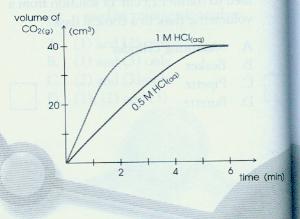
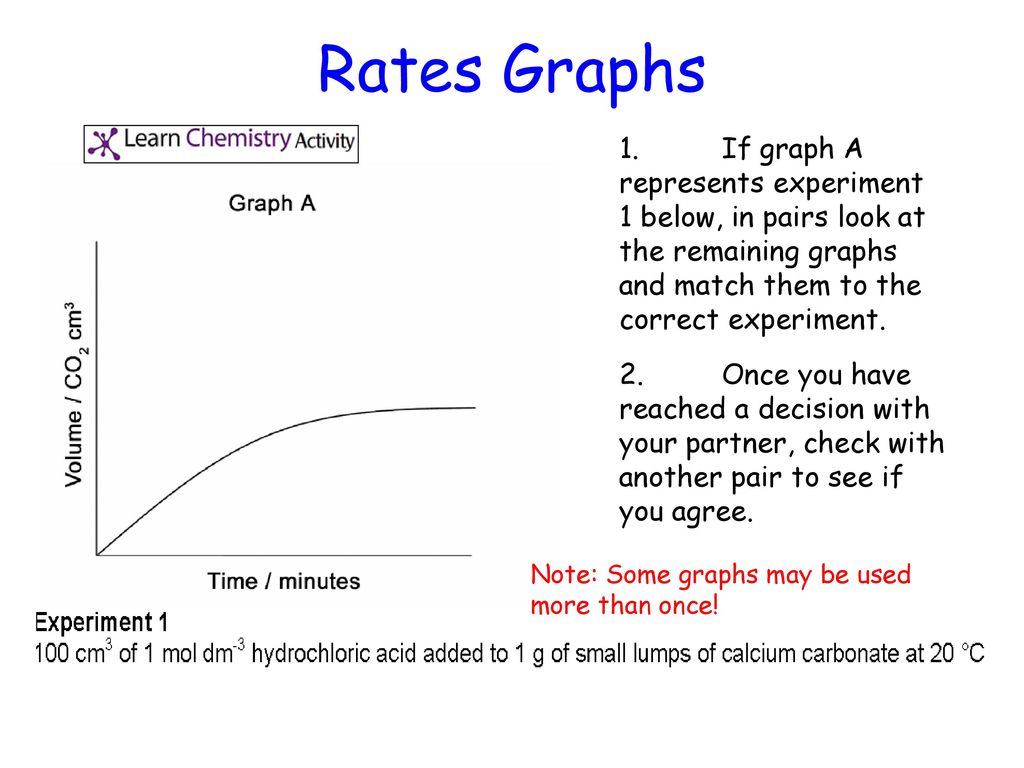
Hydrochloric acid marble chips the experiment the aim of this experiment is to find out how different variables affect the rate at which the reaction between marble chips caco and hydrochloric acid hcl takes place. The variables that i shall be changing will be the concentration of hydrochloric acid and water. There are many variables that affect. To investigate the effect of concentration on the reaction between marble chips and hydrochloric acid materials.
In the investigation i am going to find out how the surface area affects the rate of reaction by measuring the amount of gas produced and weight loss in a reaction between small large pieces of marble chips calcium carbonate and hydrochloric acid per minute. A conical flask contains the marble chips hydrochloric acid and the water that will make the reaction.
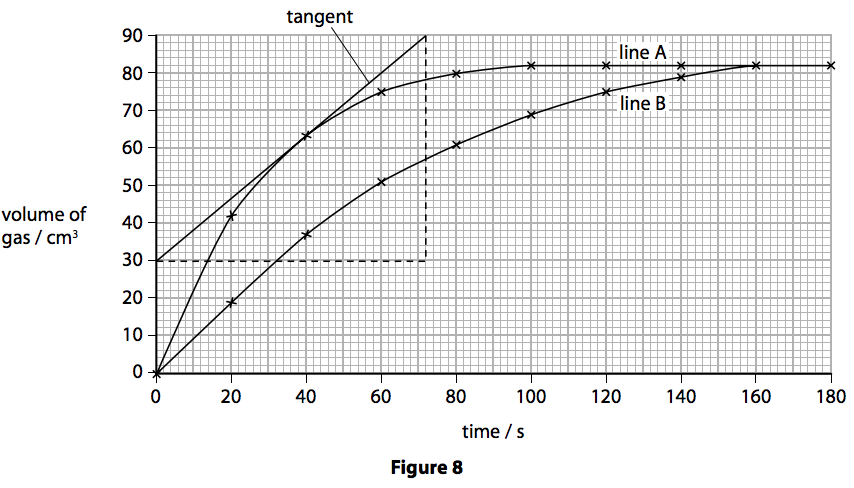
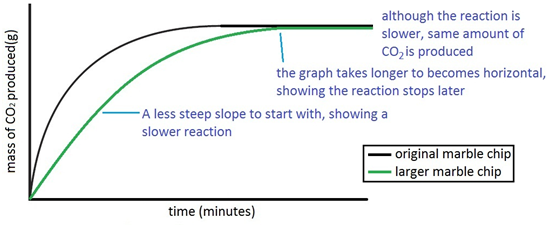










.jpg)